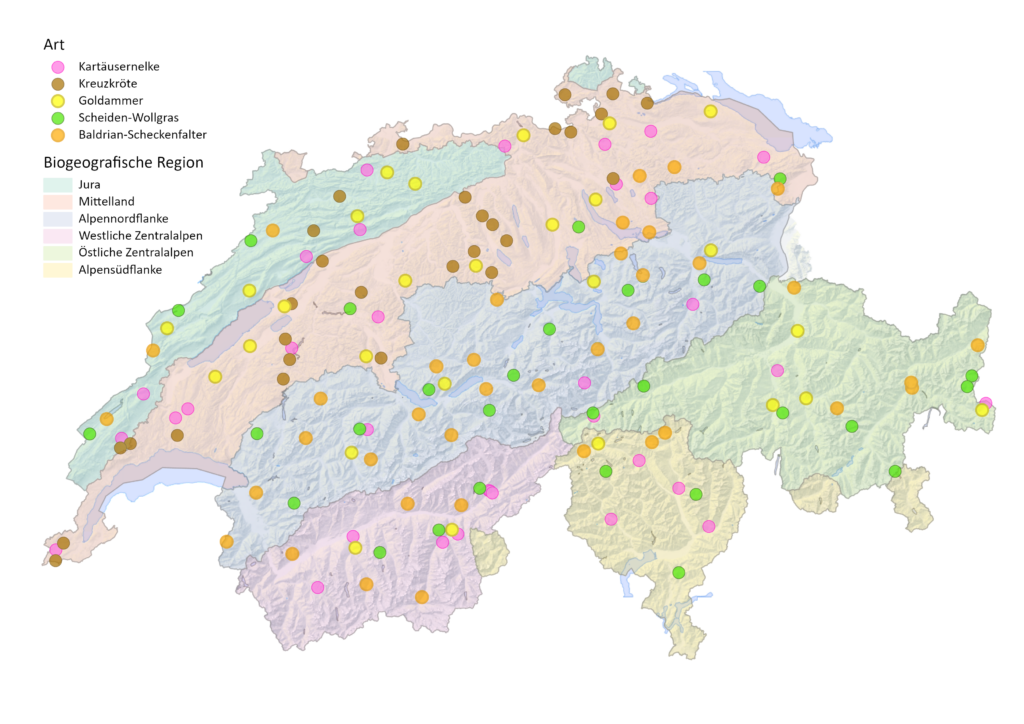
Genetic diversity is the fundamental level of biodiversity and the raw material of evolution, essential for adaptation and survival. To build capacity, knowledge and experience on how to monitor genetic diversity, we conducted a genomics-based pilot study in Switzerland. Five animal and plant species from habitats of national importance or anthropogenically modified landscapes were studied. The knowledge gained has a high potential to contribute to conservation measures that support the key action of maintaining intraspecific genetic diversity to ensure the adaptive potential of species. We are currently in the implementation phase of a genome-based monitoring programme in Switzerland.
The project is supported by the Federal Office for the Environment (FOEN) and run by the Chair of Ecological Plant Genetics in the Department of Environmental Systems Science at the Swiss Federal Institute of Technology Zurich (ETHZ), in collaboration with the Swiss Federal Institute for Forest, Snow and Landscape Research (WSL).